Making of fire works
The fireworks’ materials depends on the color that will be shown after the explosion , so they are working depending on the main components of these main materials . For instance the “Aerial shells” are consist from : a container, a lift charge, a time fuse, a burst charge, and stars/effects. The container just protects the inside contains. When the fuse enters the lowest part of the shell – which contained the lift charge -, it ignites the lift charge which create the explosion that cause the bright colors in the sky.
There are two important components:
Black powder
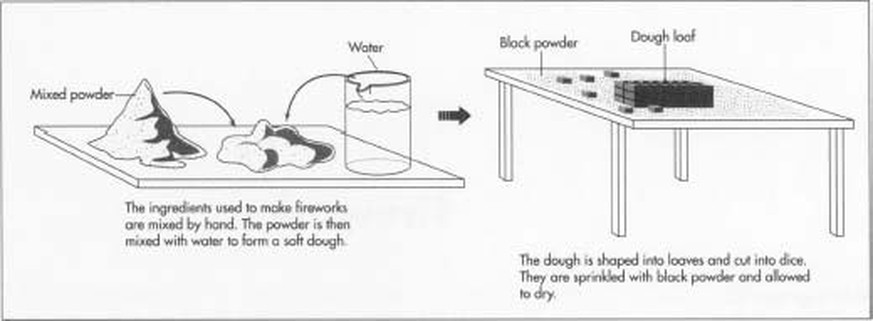
A major component of fireworks is the black powder which is a combination of about 20% charcoal, 10% sulfur and 70% potassium nitrate or sodium nitrate. Black powder burns rapidly and produces a lot of gas. If this gas is confined in a tube it reaches very high pressures. This can be used to launch a firework into the air from a « mortar » which is a tube with one open end ; like a gun or cannon. It can also be used to make a rocket by allowing the gas to escape through a small hole at one end of at tube.
Redox reaction
Redox (portmanteau for reduction-oxidation) reactions refer to all chemical reactions in which atoms have their oxidation state changed.
There are two important components:
Black powder
A major component of fireworks is the black powder which is a combination of about 20% charcoal, 10% sulfur and 70% potassium nitrate or sodium nitrate. Black powder burns rapidly and produces a lot of gas. If this gas is confined in a tube it reaches very high pressures. This can be used to launch a firework into the air from a « mortar » which is a tube with one open end ; like a gun or cannon. It can also be used to make a rocket by allowing the gas to escape through a small hole at one end of at tube.
Redox reaction
Redox (portmanteau for reduction-oxidation) reactions refer to all chemical reactions in which atoms have their oxidation state changed.
